 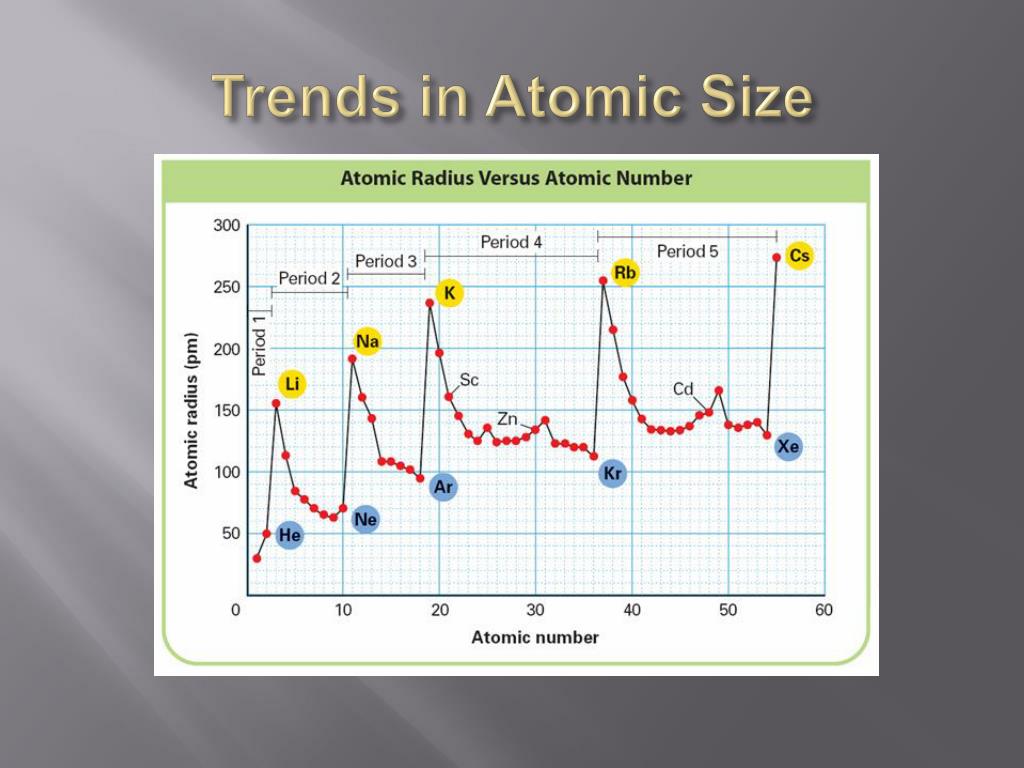
Yes, size is a factor (thus the "up" part- IE is higher in lower periods), but the observed anomaly in second-ionization energies in $\ce$ increases. Also, when you hear this incorrect explanation, you will understand what is meant. It is a little more compact to use the incorrect explanation and correct it than to explain in terms of correct. Using the common idiom: "full subshells are stable" My question is, is this approach fairly accurate or should I be looking somewhere else? In this case, Na now has reduced its size due to the fact it jumped from n=3 to n=2 level and Al also reduced in size but is still a bigger atom than Na due to our trend. Now in the case of first Ionization Energy we have: However, having Al being the smaller atom would require more energy to remove the electron from its valence shell. We see that the strongest pull towards the charged center would be Al and Na in this case. My point is, in order to get a better estimate, would it be safe to say that the Zeff charge and atom size relate to the I.E? increase up and right of the periodic table, which is not the case here. 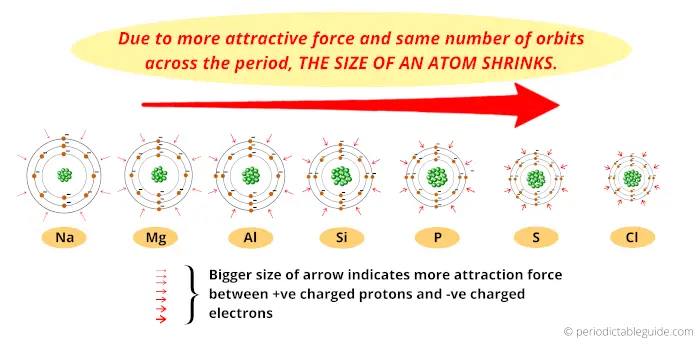
rockets up to 4562 kJ while the atoms towards the right are much lower than this. For example, Na has an I.E(1) of 495.8 kJ while its second I.E. I am familiar with the periodic trend, however things become quite different when we hit the 1st I.E. I'm trying to figure out the patterns for Ionization Energies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
